Lithium-ion batteries are widely used in various fields due to their high energy density, no memory effect, small
self-discharge and long cycle life, such as electronic products, power tools, electric vehicles, and energy storage
fields. The overall performance of a battery can be divided into two categories: electrical performance and reliability.
Life is one of the important indicators to measure its electrical performance.
For energy-type batteries, it is generally considered that when the available capacity of the battery decays to 80%
of the initial capacity, it is the end of life. The life of a battery includes cycle life and calendar life. The former refers
to the number of cycles the battery is cycled to the end of life under a certain charge and discharge system, and the
latter refers to the time required for the battery to be stored in a certain state to the end of life.
Many complex physical and chemical reactions occur during charging and discharging of lithium batteries, so there
are many factors that affect the cycle life of lithium batteries. On the other hand, cycle life testing is often
time-consuming and costly. The correct evaluation of battery life has a certain guiding role in the production and
development of lithium batteries and battery health management systems.
1. Influencing factors of cycle life
1 Aging and decay of battery materials
The materials inside lithium batteries mainly include: positive and negative active materials, binders, conductive
agents, current collectors, separators, and electrolytes. During the use of lithium batteries, these materials will be
accompanied by a certain degree of degradation and aging. Tang Zhiyuan and others believe that the capacity
decay factors of lithium manganate batteries include: the dissolution of the positive electrode material, the phase
change of the electrode material, the decomposition of the electrolyte, the formation of the interface film, and the
corrosion of the current collector.
Vetter et al. carried out a systematic and in-depth analysis on the changing mechanism of the positive electrode,
negative electrode and electrolyte of the battery in the cycle. The author believes that the formation and subsequent
growth of the negative electrode SEI film will be accompanied by the irreversible loss of active lithium, and that the
SEI film does not have a real solid electrolyte function. In addition to lithium ions, the diffusion and migration of
other substances will cause gas generation and particle rupture. In addition, the change in the volume of the
material and the precipitation of metallic lithium during the cycle will also cause a loss of capacity. The influence
on the aging and decay of the cathode material is shown in Figure 1.
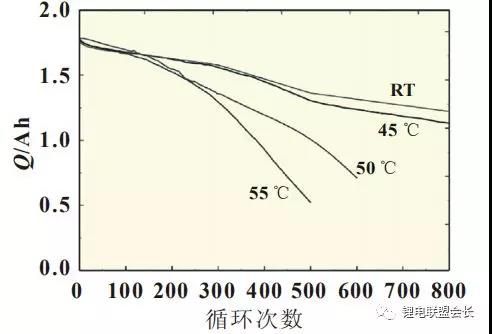
Figure 1 Aging and decay mechanism of cathode materials
Aurbach et al. disassembled the positive and negative pole pieces of the lithium cobalt oxide battery after cycling
at 25 and 40°C. The test results of SEM, XRD and FTIR showed that the positive and negative active materials were
all lost. Li Yang et al. analyzed the electrical performance of lithium iron phosphate power batteries that were cycled
6000 times and found that the capacity retention rate was 84.87%, the AC internal resistance increased by 18.25%,
and the DC internal resistance increased by 66%. The author disassembled the battery after cycling and performed
button cell performance test and SEM analysis. It was found that the performance of the negative electrode material
decayed faster after cycling, and that the expansion of the negative electrode volume and the thickening of the SEI
film were the main influencing factors.
2 Charging and discharging system
The charging and discharging system mainly includes three aspects: charging and discharging mode, rate and
cut-off conditions. In terms of charging methods, American scientist Ma Si once put forward the concept of the
best charging curve. He believes that the best charging current of the battery gradually decreases with the extension
of the charging time: I=I0e-αt. In the formula: I is the receivable charging current; I0 is the maximum initial current at
t=0; t is the charging time; α is the attenuation constant. The relationship curve between I and t is shown in Fig.2.
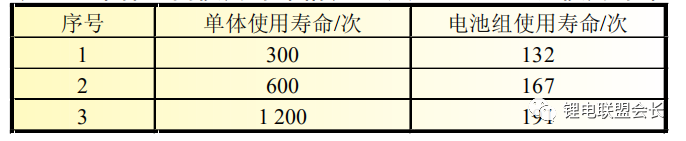
Figure 2 The battery can receive the charging current curve
In Figure 2, the lower part of the curve is the rechargeable area. Charging in this area will not cause damage to the
battery. If the charging current exceeds this area, the polarization will increase, which will not only not improve the
charging efficiency, but will also cause serious gas out of the battery and shorten the battery. Battery Life. At present,
most of the research on charging methods is carried out based on the Mars theory, that is, to make the charging
current as close to the curve as possible.
He Qiusheng and others made a comprehensive comparison of several common charging methods and found that
the constant current charging in the later stage will cause the battery to gas out due to the excessive current, which
will damage the battery; while the constant voltage charging will directly damage the battery if the current is too
large in the initial stage of charging. ; Constant current and constant voltage charging and stepped constant current
charging overcome the shortcomings of constant current charging and constant voltage charging, and are currently
widely used; reverse pulse charging can effectively eliminate polarization, but has a certain impact on life.
The charge-discharge rate and cut-off conditions also have a great influence on the battery cycle life. Li Yan et al.
studied the cycle performance of 18650 lithium cobalt oxide batteries at different discharge rates, and found that
the capacity loss rates after cycling at 0.5C, 1C and 2C discharge rates for 300 weeks were 10.5%, 14.2% and 18.8%,
respectively. And through analysis, it is concluded that the change in the structure of the positive electrode material
and the thickening of the negative electrode surface film will cause the decrease of the number of lithium ions and
the blockage of the diffusion channel, which will cause the battery capacity to decline.
K. Maher et al. increased the charge cut-off voltage of lithium cobalt oxide batteries from 4.2V to 4.9V, and found
that the structure of the electrode material has changed by testing the entropy curve of different SOCs of the
electrode after charging.
3 temperature
Different types of lithium batteries have different optimal operating temperatures, and too high or too low
temperatures will affect the service life of the battery. Ramadass et al. reported the influence of temperature on the
cycle performance of Sony 18650 lithium cobalt oxide batteries. The study found that when the test temperature
exceeds 50°C, the battery decays significantly faster than room temperature and 45°C (Figure 3), and the capacity at
high temperatures is attenuated. Attributable to the decomposition and regeneration of the negative electrode SEI
film of the battery, the loss of active lithium and the increase of the negative electrode impedance.

Figure 3 18650 battery discharge capacity curve with the number of cycles at different temperatures
Song Haishen et al. compared the electrical performance of 18650 lithium iron phosphate/graphite power batteries
at different temperatures, and obtained similar results: cycling at room temperature, the capacity of the battery
decays slowly, while under high temperature conditions of 55 and 65 ℃, The battery exhibits rapid failure behavior.
The author believes that the trace iron deposited on the graphite anode will catalyze the formation of its interface
film and have a certain influence on the capacity decay.
Zhang et al. studied the performance of lithium batteries at low temperatures and found that when the temperature
is lower than -10°C, the capacity of the battery decays sharply, and analyzed the reason for the poor
low-temperature performance. In addition to the decrease in the ionic conductivity of the electrolyte, it is also
related to the electrode material. . The author compared the EIS curves of the full battery and the positive and
negative symmetric electrodes with temperature, and found that when the temperature is lower than -10°C, the
impedance of the full battery and the half battery has an upward trend, especially the charge transfer impedance
will rise sharply. And occupy a dominant position.
4 monomer consistency
The battery pack generally connects hundreds or thousands of single cells in series and parallel. In addition to the
above-mentioned influencing factors, the cycle life of the battery pack is another important factor. Due to
differences in materials and manufacturing processes, it is difficult to guarantee the consistency of lithium batteries.
In terms of materials, the uniformity of the positive and negative materials and the electrolyte is very important. The
consistency of lithium batteries produced with the same material and in the same batch is often relatively good. In
terms of manufacturing, the production process of lithium batteries is very complicated, and each step involves
multiple process parameters. If the control is not good, it will lead to inconsistencies in parameters such as battery
voltage, capacity, and internal resistance.
Wang Zhenpo and others studied the impact of cell inconsistency on the service life of the battery pack. They
believed that the life of the battery pack is always shorter than the life of the shortest-lived single battery. A
single battery with a lifespan of 1,000 times has a lifespan of less than 200 after being assembled. Second, and
the increase in battery life is disproportionate to the increase in battery life (Table 1).
Table 1 Theoretical service life of power battery pack under different service life of monomer

Based on Thevenin equivalent circuit, Chen Qiang and others investigated the impact of the ohmic resistance,
capacity and polarization differences of single cells on the performance of the series battery pack, and found that
the capacity difference has the greatest impact.
Before the battery is actually used in groups, it will go through the process of screening and matching to eliminate
the monomers with large differences in performance parameters, and minimize the impact of the differences in the
battery manufacturing process on the performance. Batteries are generally grouped according to parameters such
as battery capacity, voltage, internal resistance, and self-discharge. However, rapid detection of battery
self-discharge is a research difficulty. The self-discharge of the single battery will cause the SOC of each battery in
the battery pack to be inconsistent, and affect the capacity of the entire battery pack. Generally speaking, the higher
the temperature, the greater the self-discharge of the battery. If the design of the battery pack box is unreasonable,
the internal resistance and self-discharge degree of the batteries in different positions will be affected to a certain
extent due to the difference in heat dissipation.
2. Cycle life prediction
Due to the long time and high cost of battery cycle life testing, the establishment of life model and life evaluation
and prediction have become research hotspots of domestic and foreign scholars. Lithium battery life prediction
methods can be divided into three categories according to information sources: prediction based on capacity
decline mechanism, prediction based on characteristic parameters, and prediction based on data-driven.
1 Forecast based on capacity decline mechanism
Mechanism-based prediction is based on the battery's internal structure and material aging and decay mechanism
during cycling to estimate the battery's life. This method requires the use of basic models to describe the physical
and chemical reaction processes that occur inside the battery, such as Ohm's law, electrochemical polarization,
concentration polarization, and internal diffusion of electrode materials.
Based on the loss of active lithium during the battery cycle, Ning et al. used first principles to simulate the capacity
decline model of lithium cobalt oxide batteries. The influencing parameters include exchange current density, DOD,
interface membrane impedance, and charge cut-off voltage. The author compares the life prediction model with the
measured data and finds that the model is very close to the actual test results.
Virkar proposed a battery degradation model based on non-equilibrium thermodynamics, taking into account the
influence of chemical potential and SEI film on capacity degradation, and pointed out that there will be unbalanced
monomers in the series battery pack, and the interface between the positive electrode and the electrolyte SEI film
may also be produced, leading to increased capacity degradation.
2 Prediction based on characteristic parameters
Prediction based on characteristic parameters refers to the use of changes in certain characteristic factors of the
battery during the aging process to predict battery life. At present, researchers pay most attention to the relationship
between EIS and cycle life. Li et al. studied the changes in impedance spectra of commercial lithium cobalt oxide
batteries during 1C charge and discharge cycles, and observed the changes in electrode materials by XRD, TEM and
SEM. They found that the Nyquist curves of the positive and negative electrodes of lithium batteries correspond to
The size of the semicircle in the low frequency region of the interfacial membrane impedance increases with the
increase of the number of cycles. Based on this, the cycle life of the battery can be inferred.
EIS can give a finer description of battery impedance, but the test instrument is susceptible to external interference
and it is difficult to effectively analyze complex spectra. In contrast, the measurement of pulse impedance is simple
and easy, and online monitoring can be quickly realized.
3 Based on data-driven forecasting
The data-driven method refers to the direct analysis of test data to mine the law without considering the physical
and chemical reactions and mechanisms inside the battery. It is a simulation method based on experience. The
more common ones are time series model (AR), artificial neural network model (ANN) and correlation vector
method (RVM).
The AR model is based on the measured data at some previous time points to infer the predicted value in the
current state, and has linear characteristics. Taking into account the non-linear relationship between battery capacity
attenuation and the number of cycles, Luo Yue proposed an improved non-linear AR model, which introduced an
accelerated degradation factor in the later stage of the prediction to improve the accuracy of the prediction.
The ANN model is an artificial intelligence network system composed of multiple neurons according to certain rules,
and is a typical nonlinear model. The RVM model is a data regression analysis method, which can flexibly control
over-fitting and under-fitting by adjusting parameters, and has the characteristics of probabilistic prediction. The
prediction method based on the internal mechanism has better theoretical support and better accuracy, but the
complexity is large. The advantage of the data-driven method is that it is simple and practical, but because the
acquired data cannot cover all parameters, it also has certain limitations. sex.
Three, concluding remarks
This article mainly introduces the influencing factors of power lithium-ion battery cycle life and the research of life
prediction model. It can be seen that there are many factors that affect the cycle life of power lithium batteries, and
for lithium batteries of different materials and structures, the influencing factors are also different.
From the analysis in the article, we can see that we can extend the battery life by controlling the parameters, such as
allowing the battery to work under the appropriate temperature, rate, and charging and discharging conditions.
Relatively speaking, the influencing factors of the battery pack’s cycle life are more complicated, because these
factors will have mutual coupling, and the monomer consistency problem will cause the battery pack’s
performance to not be fully utilized, and severely shorten the battery pack’s cycle. life.
When predicting the cycle life of the battery, it can be based on the internal mechanism of the battery, a certain
characteristic parameter or a large amount of data that has been measured, and the establishment of an accurate,
reasonable and simple and operable model can accurately evaluate the cycle life of the battery and further optimize
its performance. Has important meaning.





























